Molecular Level
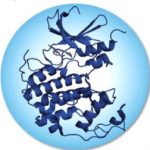
Plant protein transporters function to acquire nutrients from the soil, and undergo conformational changes when transferring substrate, such as nitrate ion (NO3-) from soil to cell. The recently obtained crystal structure of a key NO3- transporter in Arabidopsis (AtNRT1.1) has provided a first glimpse of the complex molecular interplay between transporter activity and post-translational modifications.
However, the mechanistic basis of NO3- uptake and sensing still remains elusive. In principle, in silico approaches such as Molecular Dynamics (MD) simulation can help determine this mechanism by filling gaps in knowledge about receptor phosphorylation, dynamic receptor dimerization, and post-translational modifications. We are currently performing large-scale atomistic simulations of AtNRT1.1 molecular model. The inputs required for these simulations include environmental factors such as temperature, NO3-level, and cellular pressure and salt concentration. One output from this model will be a predicted strategy to manipulate NO3- uptake via point mutations, resulting in enhanced NO3--uptake efficiency. The predicted change in NOO3- uptake rate will then be used as an input for subsequent modeling levels.

